Science and technology has rapidly evolved over the last century. A lot of serious diseases/illnesses have been stemmed and minimized with the help of the innovations in medical sciences. If you think about it, it’s amazing the lengths the capacity of human knowledge has reached and achieved! Covid-19, however, has shown us how far we still have to go in terms of invention and innovation.
In light of the pandemic, there are some discussions about some people being hesitant with the legitimacy of medical sciences as we know it. What we don’t know is the meticulous and tedious process our medical devices go through to verify its authenticity. In this article, we provide you with free and ready-made samples of Clinical Evaluation Report for your perusal. Keep on reading to find out more!
10+ Clinical Evaluation Report Samples
1. Clinical Evaluation Report
2. Clinical Evaluation Assessment Report
3. Clinical Evaluation Report and Plan
4. Customer Clinical Evaluation Report
5. Clinical Evaluation Report Sample
6. Clinical Evaluation Summary Report
7. Medical Devices Clinical Evaluation Report
8. Model Clinical Evaluation Report
9. Standard Clinical Evaluation Report
10. Editable Clinical Evaluation Report
11. Clinical Evaluation Organization Report
What Is a Clinical Evaluation Report?
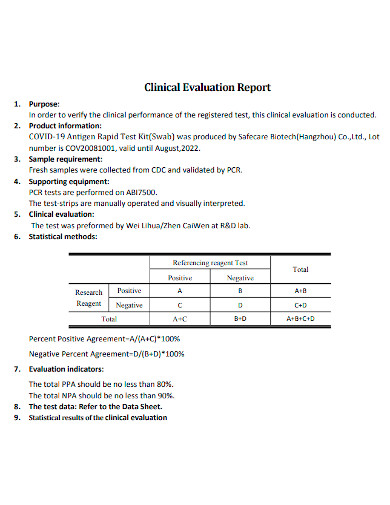
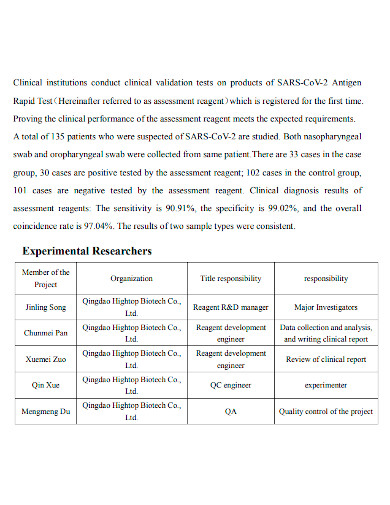
A Clinical Evaluation is a method for gathering and analyzing all clinical data related to a device in order to determine whether there is sufficient clinical evidence to establish compliance with regulatory standards. A Clinical Evaluation Report (CER) therefore documents the procedure and its outcomes. It is made up of evaluated clinical data from either a clinical examination of the medical device device or the results of other studies on devices that are substantially equivalent to the said device. The CER certifies that the certain technology accomplishes its goal while causing no additional danger to potential users or patients. A clinical performance and clinical safety analysis should be included in the report.
Only by analyzing the entire collection of accessible data will scientists be able to choose the simplest, safest, and most effective clinical trial configuration. There is a need to think very carefully about what aspects of safety and performance need to be tested in humans, and which can be supported by pre-clinical research, from both an ethical and a business standpoint.
How to Make a Clinical Evaluation Report?
If you have not written a Clinical Evaluation report before, you presumably might be wondering the details and resources needed to complete one. The depth of your report actually depends on the medical device; the higher the potential risks it might have, the more detailed it should be. If you’re interested in writing one on your own, here are the fundamental steps in the report’s structure to guide you:
1. The general information
The first step is to indicate the device and the manufacturer name. This section is intended to be a thorough examination of the devices addressed in the CER, encompassing both technical and clinical issues. It should clearly describe how the device operates as well as the technology it employs. Items to include should be a picture or diagram of the device, classify the device by group, give a brief physical and chemical description of the device and its components, and specify whether the device works with drugs, tissues, or blood.
2. The overview
This part of the report is an extensive summary of the Clinical Evaluation. This is intended for agencies and notified bodies outside the field. Items to include here are the benefits and risk of the product, the product’s intended target users, and its significance in the medical sciences.
3. The clinical background
This portion of the CER should primarily consist of a research-based overview of the clinical environment in which the medical device is used. In this section, offer a synopsis of the literature review to let reviewers visualize the current state of the art for the medical device under consideration.
4. Conclusion
For the last section of the report, it should essentially be a statement that justifies the device’s compliance with critical patient safety regulations. It should also be indicated whether the gadget, as well as any hazards linked with it, is better than medical alternative.
FAQs
How many phases are in a clinical trial?
Each clinical trial goes through three stages before being approved by the Food and Drug Administration (FDA).
What is clinical data in healthcare?
Clinical data is information obtained from patient care to clinical research level for the goal of clinical research: broad applications within a health system.
What is the use of clinical data?
It includes information on determinants of health, health metrics, and health status, as well as documentation of care delivery. These records are collected for a variety of reasons and stored in a variety of databases throughout the healthcare system.
All in all, it is vital for medical devices to undergo a process of thorough investigation in order to verify its legitimacy in curing or helping those in need. There have been a lot of breakthroughs in the medical sciences over the years, which is why this report is more relevant than ever. With the use of our free, ready-made, and downloadable Clinical Evaluation Report, you can write one up without hassle! Print these out and you’re good to go.
Related Posts
FREE 10+ Sample Job Evaluation Forms
FREE 10+ Sample Presentation Evaluation Forms
FREE 10+ Supervisor Evaluation Samples
FREE 10+ Patient Feedback Forms
FREE 9+ Sample Medical Necessity Forms
FREE 9+ Medical Form Samples
FREE 8+ Against Medical Advice Forms
FREE 7+ Evaluation Plan Templates
FREE 6+ Sample Supplier Evaluation Forms
FREE 5+ Charity Impact Report Samples
FREE 44+ Assessment Forms
FREE 21+ Employee Evaluation Form Samples
6+ Sample Nursing Assessment Forms
FREE 10+ Sample Self Evaluation Forms
FREE 6+ Sample Presentations