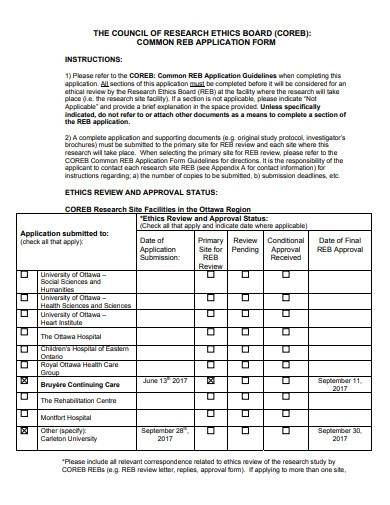
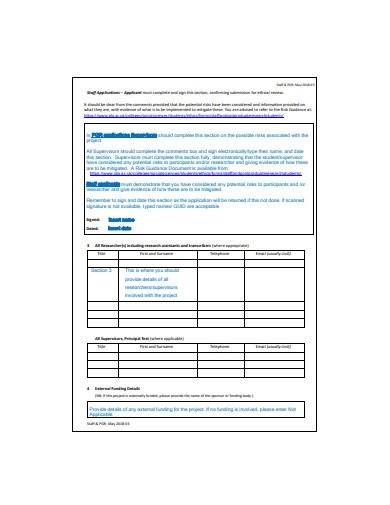
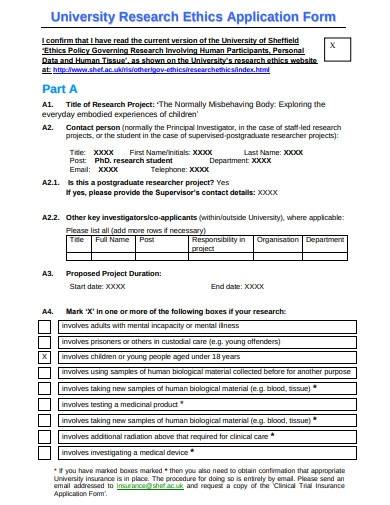
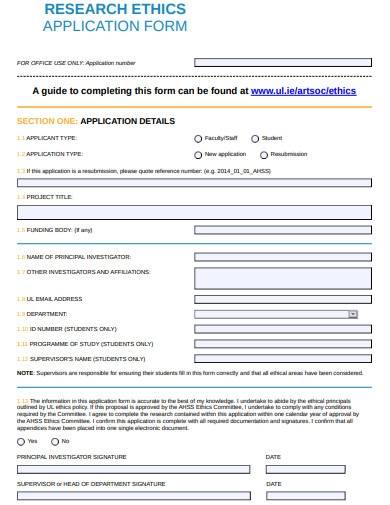
The purpose of the form is to ensure that any qualiatative research or research project proposal is conducted in an ethical statement and responsible manner, and that the waiver of moral rights, safety policy, and well-being of participants are protected. The form typically includes information about the research study, such as the research question, methodology, participant recruitment, and sample data analysis procedures. It also includes information about how the researcher plans to obtain informed consent from participants and how they plan to protect their confidentiality.
FREE 10+ Research Ethics Form Samples & Templates
1. Research Ethics Application Form
2. Research Ethics Form Template
3. Safeguard Referral Form Template
4. Common Reb Application Form
5. Research Ethics Approval Proposal Form
6. Sample Research Ethics Form
7. Research Ethics Review Form
8. Formal Research Ethics Form
9. University Research Ethics Application Form
10. Ethics Research Approval Form
11. Standard Research Ethics Application Form
What is Research Ethics Form?
A research ethics form, also known as an ethics application form, is a document used by researchers to obtain approval from an ethics committee or Institutional Review Board (IRB) before conducting a research study that involves human subjects.
How To Make Research Ethics Form?
The ethics committee or IRB will review the form to determine whether the research study meets ethical standards and whether it is safe and appropriate to conduct the study. The committee may request modifications to the study design or procedures in order to ensure that the study is conducted in an ethical and responsible manner. If the committee approves the study, the researcher can proceed with the research. The process of making a research ethics form may vary depending on the institution and the type of research being conducted. However, here are some general steps that can guide you in creating a research ethics form:
Step 1- Determine Ethical Guidelines
Familiarize yourself with the ethical guidelines and regulations that apply to your research. This could include international, national, and institutional ethical guidelines and regulations. Then, Identify the key elements of your research study, such as the research question, study design, data collection and analysis methods, and proposal for recruitment samples.
Step 2- Consider Issues and Risks
Consider the potential ethical issues and risks associated with your research. This could include issues such as confidentiality, informed consent, and potential harm to participants. Develop a clear and detailed informed consent form that includes information about the study, participant rights, and potential risks and benefits.
Step 3- Create Data Management Plan
Create a data management plan that outlines how you plan to manage and store the data collected during the study. Using the information gathered above, draft the research ethics form that includes all the relevant information required by the ethics committee or Institutional Review Board (IRB). Ensure that the form is clear and easy to understand.
Step 4- Submit Form
Submit the research ethics form to the ethics committee or IRB for review and approval. The ethics committee or IRB may request modifications to the research ethics form or study design. If this happens, revise the form and resubmit it for further review.
How long does it take for a research ethics form to be approved?
The time it takes for a research ethics form to be approved can vary depending on the complexity of the study and the workload of the ethics committee or IRB. It is important to plan ahead and allow sufficient time for the review process.
What happens if the research ethics form is not approved?
If the research ethics form is not approved, the researcher may need to modify the study design or procedures to ensure that the study is conducted ethically and responsibly. The researcher may also need to resubmit the form for further review.
What are some common ethical issues in research?
Some common ethical issues in research include confidentiality, informed consent, potential harm to participants, and conflicts of interest. It is important to consider these issues when designing a research study plan and to address them in the research ethics samples.
A research ethics form is important because it ensures that the study is conducted in an ethical and responsible manner, and that the rights, welfare, and safety of the participants are protected.
Related Posts
FREE 13+ Sample Service Hour Forms in PDF MS Word
FREE 8+ Sample Internship Recommendation Letter Templates in ...
Sample Unsolicited Proposal Template
FREE 20+ Research Paper Outlines in PDF MS Word
FREE 50+ Sample Medical Forms in PDF MS Word
FREE 35+ Proposal Letter Format Samples in PDF MS Word ...
Sample Committee Report Template
FREE 12+ Research Proposal Samples in PDF MS Word | Pages
FREE 38+ Sample Recommendation Letter Templates in PDF
FREE 8+ Sample Accounting Memo Templates in MS Word PDF
FREE 6+ Sample APA Format Templates in PDF MS Word
FREE 6+ Sample Business Case Analysis Templates in PDF MS ...
FREE 7+ Program Evaluation Forms in PDF MS Word
FREE 8+ Sample Professional Proposal Templates in PDF MS ...
FREE 36+ Sample White Paper Templates in PDF